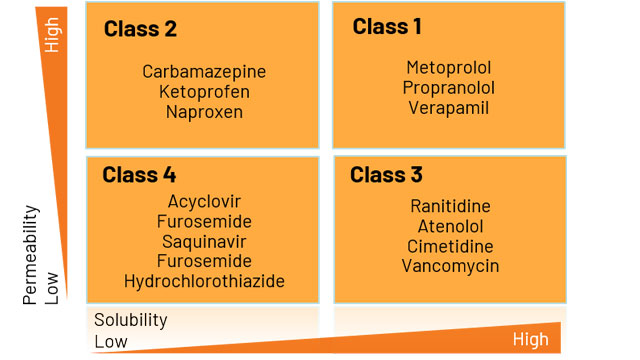
The BCS classification system is based on the scientific rationale that, if the highest dose of a drug candidate is readily soluble in the average fluid volume present in the stomach (250 ml) and the drug is more than >85% absorbed, then the in vitro drug product dissolution profiles should allow assessment of the equivalence of different drug formulations. Bcs Classification Of Drugs List Fda Bcs Classification List Of DrugsĪ waiver of In-vivo Bioavailability and Biioequivalence studies based on the BCS classification can therefore save pharmaceutical companies a significant amount of development time and reduce development costs. Guideline On The Investigation Of Bioequivalence (European Medicines Agency, London, 20 January 2010, CPMP/EWP/QWP/1401/98 Rev. January 2013 Adopted and adapted mainly from the following: 1. GUIDANCE ON BIOPHARMACEUTICS CLASSIFICATION SYSTEM (BCS)-BASED BIOWAIVER National Pharmaceutical Control Bureau, Ministry Of Health Malaysia. BCS-based biowaivers are applicable to drug products where the drug substance or substances exhibit high solubility and, either high permeability (BCS Class I) or low permeability (BCS Class III). We evaluated 263 approved generic drugs of IR products listed on the WHO EML to find out the distribution of BCS Class 1, 2, 3, and 4 drugs in approved ANDA applications. Keywords: Silica lipid, Bioavailability, Permeability, Microparticles. BCS classification (Biopharmaceutics Classification System) is classified into four main classes which categorized the drugs on major bioavailability parameters.

The objective of this study was to use the World Health Organization Model List of Essential Medicines to determine the distribution of BCS Class 1, 2, 3, and 4 drugs. Biopharmaceutics Classification System (BCS) has provided a mechanistic framework for understanding the concept of drug absorption in terms of permeability and solubility. bioavailability of BCS class IV drugs effectively. The Biopharmaceutics Classification system (BCS) classifies drug substances based on aqueous solubility and intestinal permeability. The principles of the BCS classification system can be applied to NDA and ANDA approvals as well as to scale-up and post approval changes in drug manufacturing. A drug product is eligible for a BCS-based biowaiver provid that the drug substance(s) satisfy the ed criteria regarding solubility and permeability (BCS Class I and III), the drug product is an immediate-release oral dosage form with systemic action, and the drug product is dosage the same form and strength as the reference product. Nevertheless, the HEC has evolved a procedure whereby it is able to meet these challenges and address all the complexities involved.The “” is an FDA guidance document, which allows pharmaceutical companies to forego clinical bioequivalence studies, if their drug product meets the specification detailed in the guidance. With the proliferation of degree-awarding institutions – both in the private and public sectors – and given the diversity of taught programs and disciplines, the comparability and method of recognition for degrees has become more confusing In such a scenario, the equivalence of degrees and diplomas and their recognition pose complex issues for educational authorities. The accreditation of institutions, courses and degree programs has become increasingly challenging with increasing international collaboration in the fields of science and technology, as well as the increased mobility of students and scholars in the modern world. Poor oral bioavailability may be due to poor aqueous. HEC revised the rates of equivalence fee for Local Degree/Deeni Asnads w.e.f. BCS class 4 drugs are having poor oral bioavailability due to poor solubility and poor permeability.
#Bcs class 4 drugs list manual#
Applicants are advised to apply for equivalence of their degrees at as no manual applications for equivalence of Pakistani or Foreign degrees will be accepted henceforth.Īdvisory for Admission in Foreign Medical Universities/Institutions in the present study we have selected one such bcs class iv drug, hydrochlorothiazide (hctz) 7 which is a well categorized thiazide diuretic, considered as the first-line of treatment for hypertension and listed as an essential medicine in who list. Higher Education Commission has launched online portal for equivalence of degrees.
